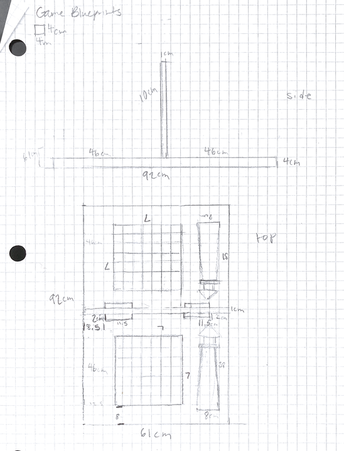
In Chemistry we had to build a board game with our groups. The game had have chemical and physical changes involved. The game had to show six reactions occurring: a single and double replacement, a production of a gas, have an LED light turn on, a chemical and physical change of energy.
Safety Precautions
Players: 2
Goal: Sink all of the opponent’s ships
- The game contains lead (DO NOT PUT YOUR HAND ON YOUR FACE)
- Wear gloves and wash hands after
Players: 2
Goal: Sink all of the opponent’s ships
- Each of the players places ships on the lower grid and uses the chemicals that correspond with the gridlines
- Place ships anywhere on the grid
- Once ships are set up, you will try to hit the other ships by guessing where they are on the other player’s grid
- Guess in a letter-number fashion
- The letter-number coordinates will correspond to a coordinate on the other player’s grid
- If you hit your opponent’s ship, they will say “hit”
- If you miss, they will say “miss”
- Mark the spots that you’ve guessed on the upper board with a marker
- If the other player hits any part of one of your ships (1 of 3 of the squares), the whole ship “sinks” and you must do the chemical equation written on the ship.
- Hits are “X” and misses are “O”
- Whoever sinks every one of the other player’s ships wins and gets to light the lighthouse on their lower board
Single replacement reaction
Reaction in word form: Copper Chloride + Magnesium ------> Magnesium Chloride + Copper
Reaction chemical formula: CuCl2 + Mg —> MgCl2 + Cu
We had a breadboard with an LED set up, and used alligator clips to connect magnesium to the circuit. The player would pour copper chloride onto the magnesium and create a reaction that would it.
definition: a type of chemical reaction where an element reacts with a compound and takes the place of another element in that compound
Double replacement reaction
Reaction in word form: Potassium chloride + lead nitrate ---> lead chloride + potassium nitrate
Reaction chemical formula: 2KCl + Pb(NO3)2 ------> 2KNO3 + PbCl2
The player would put a few drops of both potassium chloride and lead nitrate onto a laminated sheet, and the solution would turn yellow. The combination formed a precipitate, following the solubility guidelines.
definition: a type of chemical reaction where two compounds react and the positive and negative ions of the reactants switch places
Production of a gas
Reaction in word form: sodium bicarbonate + acetic acid ---> carbon dioxide + water + sodium ion + acetate ion
Reaction chemical formula: NaHCO3 + CH3COOH → CO2 + H2O + Na + CH3COO
The player would put a few drops of acetic acid into a small container of sodium bicarbonate, which would create bubbles.
Reflection
During the whole project I felt like a nuisance, it didn't matter if I tried to help or not. I liked painting on the board, and feeling like I was helping out. I didn't like how nothing was planned out all that well, so our group was really stressed out in the end. I think if we've made an outline of things to do on certain days, and every member had a job to get done that was due on a day earlier, or more, then when the whole project was due so we could all look it over and make last minute changes. I also didn't like feeling like an outcast with my group. It was mostly my fault because I didn't come to class sometimes.